OUR EXPERTISE
A know-how and competencies
As an independent pharmaceutical company, our strategy is mainly based on mature products with high medical values.
Our expertise is used to acquire and maintain products that have stood the test of time while continuing to meet an important medical need.
With the benefit of their extensive experience in the pharmaceutical industry, Delbert Pharma has built a multidisciplinary team eager to put their skills at the service of patients. Experts in the health field, Delbert Pharma has an expertise in regulatory affairs, quality assurance as well as in the scientific, medical and legal fields.
History of an independent French laboratory

2002 – CREATION OF LABORATOIRES DELBERT by Michel BERTHIER, pharmacist.
Experts in regulatory affairs, Michel Berthier and his daughter Laurence Delemotte, created Laboratoires DELBERT. The two pharmacists offer a service of regulatory activities: more than 300 Marketing Authorization files for generic drugs were registered in 10 years
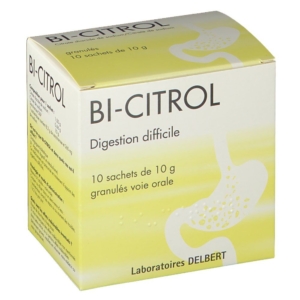
2012 – LABORATOIRES DELBERT, A PHARMACEUTICAL COMPANY
Laboratoires DELBERT acquire their first pharmaceutical specialty: Bi-citrol® and become a pharmaceutical company with an “exploitant” status.
2013 – BIRTH OF A START UP
Laboratoires DELBERT are bought by Thierry HOFFMANN, industrial pharmacist and Marc CHILDS, psychiatrist.

2014 – CREATING A MODEL
Acquisition and return to the market of Vercyte® (Pipobroman).
In a context of shortage of Vercyte® specialty (Pipobroman), Laboratoires DELBERT acquired the product. They register a new supplier of active ingredients and find a production site for the finished product capable of handling highly toxic substances.
2016 – EXPANSION OF PRODUCT’S PORTFOLIO
Acquisition of the Piportil ® / Piportil ® L4 (Pipotiazine) range
Threatened with marketing discontinuation, Laboratoires DELBERT commit to maintaining PIPORTIL and PIPORTIL L4 in the psychiatric therapeutic arsenal.
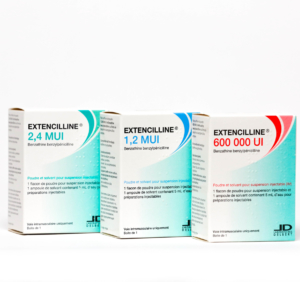
2017 – REDEVELOP EUROPEAN KNOW-HOW
Returned to the market of Extencilline® (Benzathine benzylpénicilline)
Laboratoires DELBERT identify a European production site with an infrastructure suitable for the fermentation of penicillin. Reactivation of the production site allows benzathine benzylpenicillin-based medicine to be made available on the market again.
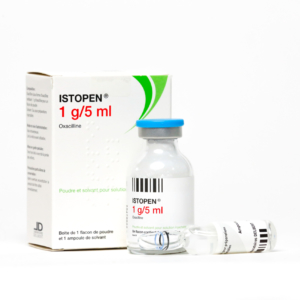
2018 – BEGINNING OF INTERNATIONALIZATION
Acquisition of the MAs for Bristopen® / Penstapho® / Prostaphlin® (Oxacillin) in France, Italy and the Czech Republic.
Acquisition of Mucomystendo® (Acétylcysteine) MAs in France
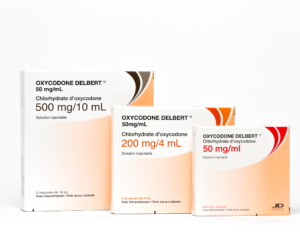
2019 – INTERNATIONALIZATION DEVELOPMENT
Launch of Oxycodone DELBERT®
Acquisition of the Marketing Authorization for Amukin® (Amikacine) and PENADUR® (benzathine benzyl penicillin) in Belgium and Luxembourg
2024 – LABORATOIRES DELBERT BECOMES DELBERT PHARMA
New commercial name: following its internationalization, Laboratoires Delbert becomes Delbert Pharma.
Delbert Pharma has built up pays particular attention to the choice of their partners in order to create a solid network and offer a flexible and scalable structure.
To this end, the company carries out different activities:
- Holding and using their own marketing authorizations
- The supply of various pharmaceutical and regulatory services for laboratories wishing to develop their activity on the French market,
- Operating AMM on behalf of third parties
- Licensing at international level
Our drugs are developed and manufactured to meet the highest standards. Marketing the best quality products is at the heart of our concerns. This is why we mainly select our partners on European production sites. The quality of these sites is based on GMP (good manufacturing practices), the European Pharmacopoeia, as well as on environmental requirements.
DELBERT PHARMA thus offers medicine in various fields such as oncology, infectious diseases, psychiatry and neurology.




